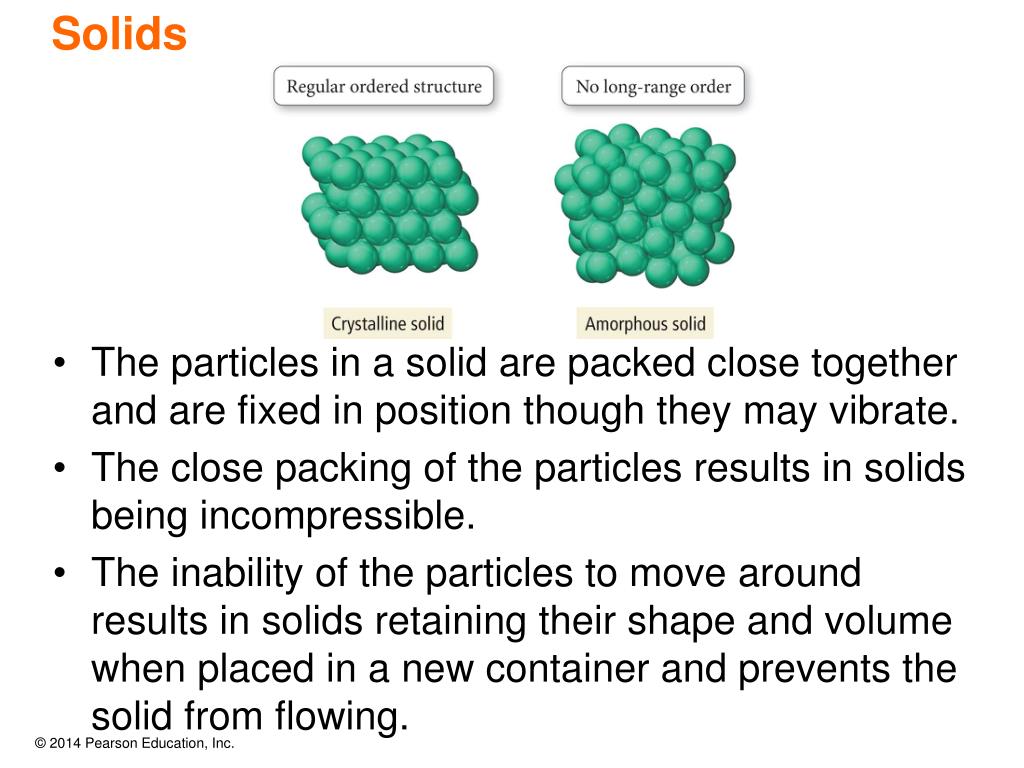
Particles move around each other, which means gases flow readily. Amorphous Solids Particles are arranged randomly. They have a regular and ordered arrangement resulting in a definite shape. In addition, there is enough free space between gas particles to compress them. Crystalline Solids Particles are arranged in a repeating pattern. Amorphous compounds melt gradually across a temperature range, whereas crystalline solids have > sharp melting points. 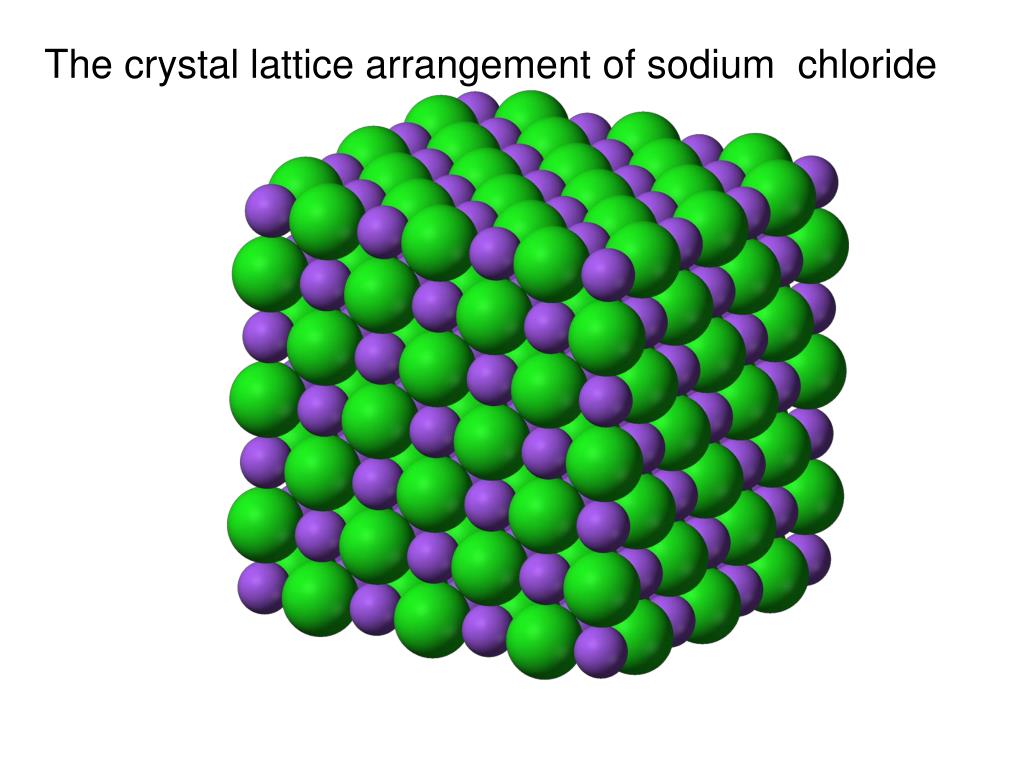
In the solid the particles vibrate in place. Their vibration is related to their kinetic energy. Melting Points: Melting points is the temperature at which substances melt. Solid In a solid, the attractive forces keep the particles together tightly enough so that the particles do not move past each other. The particles in a crystalline solid are arranged A)NH3 solid B)NH3 liquid C)NH3 gas. In contrast to solids, gases also fill the shapes of their containers. A crystal is a substance in which the particles are arranged in an orderly, repeating, three-dimensional pattern. As a result, crystalline materials have long-range order, whereas amorphous solids only have short-range order. Any substance which has mass and occupies space made up of atoms. Crystal has a SPECIFIC GEOMETRY SHAPE WITH DEFINITE EDGE Crystalline solid cleavage. It is because of this that solids and liquids are sometimes called condensed phases. Crystalline Solid has an orderly arrangement of their constituent particle. In crystalline solids the constituent particles are arranged in a regular and peri- odic pattern and give a well defined shape to it. One similarity between solid and liquid particles is that it is not possible to compress either of them. Solids in which particles are arranged in a repeating, three-dimensional pattern are called crystals or crystalline solids. In addition, this allows them to adapt to the shape of a container, whereas solids cannot. For example, liquid particles have fluidity, which means it is possible to make a liquid flow. Solid particles vary considerably from liquids. As solid particles are unable to move past each other, it is not easy to make a solid flow.Īlthough gases, liquids and solids feature atoms, molecules and sometimes ions as their key components, they have significant microscopic differences. It is not easy to compress a solid, as there is not much space between the particles. A solid’s volume and shape are fixed, which means the particles are rigid and stay in place.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
